Introduction
A diagnosis of a congenital heart disease was more of an anatomical finding than a clinical condition in the past. Until the beginning of the 20th century, congenital heart disorders had been rarely recognized in live individuals and even heart atlases were addressed rather to pathologists than clinicians (Freedom, Yoo, Mikailian, Williams, 2003). The scientific achievements of the mid and especially the late 20th century revolutionized pediatric cardiology. Now, most, if not all, heart defects can be diagnosed and effectively managed in both children and adults.
 Get a price quote
Get a price quote
- Free bibliography page
- Free title page
- Free formatting (APA, MLA, Harvard, Chicago/Turabian)
- Free revision (within 2 days)
- Free from AI-written content
- 24/7 support
- Approx. 300 words/page
- Font: 12 point Times New Roman
- Double and single spacing
- 1 inch margin
- Up-to-date sources
- Any citation style
A ventricular septal defect is the most common congenital heart disorder. It has been studied so deeply that some authors claim today that the ventricular septal defect (VSD) is a ‘simple’ disorder. VSD diagnostics nowadays is non-invasive, the treatment of this condition typically carries minimal risks. Morbidity and mortality from a VSD approach is zero in current treatment protocols. The expected life duration can now be almost the same as for the general population.
Epidemiology
The ventricular septal defect is the most common of significant congenital heart diseases. Some reports suggest its prevalence is as high as 25.2 per 1,000 births, probably when small muscular defects, which typically close spontaneously in the first year of life, are included (Freedom et al., 2004). Most papers agree the incidence is around 0.3 to 3.3 cases per 1,000 live births, more prevalent in female (Freedom et al., 2004; Ramaswamy, 2013).
Anatomy
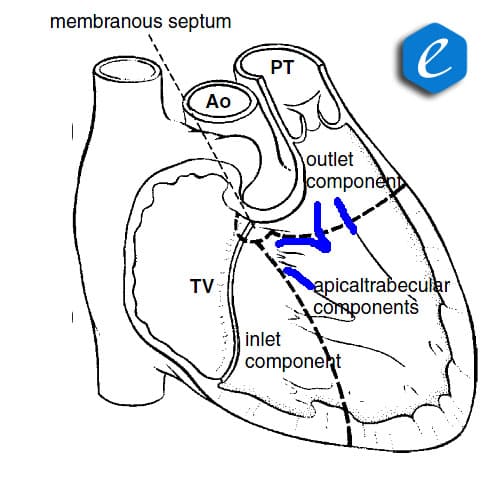
The interventricular septum, the division between the right and left ventricles, has complex morphology. Normally, the interventricluar septum has three portions:
- Inlet portion under the tricuspid valve.
- Outlet portion under the pulmonary valve.
- Trabecular portion between them, this part extends to the apex of the heart.
All three portions are muscular in origin and come up against the membranous firm partition (the membranous septum) at the crest of the heart (Fig. 1).
Figure 1. Normal anatomy of the interventricular septum. Inlet, outlet, and trabecular components are muscular, the membranous septum is fibrous. The trabecular portion gives rise to a muscular band, called the moderator band, which divides into two limbs superiorly (marked blue) (Stark, de Leval, Tsang, 2006).
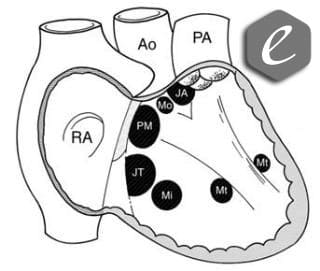
Ventricular septal defects can occur anywhere within the ventricular septum. Thus, over a dozen of anatomical classifications have been proposed to describe the VSD. In general, the classification systems attribute the VSD to the corresponding atrioventricular valves, arterial valves, the conduction axis, or the locus of the ventricular septum itself (Freedom et al., 2004). The most common type of the VSD is perimembranous indicating its location around the membranous septum or within it. Juxta-tricuspid defects do not extend to the membranous septum but develop under the tricuspid valve. Juxta-arterial defects do not contact with the membranous septum and support the pulmonary valve. Muscular defects do not touch membranous septum or the valves. Muscular defects may correspond to the inlet portion (muscular inlet), chain to the moderator band (muscular trabecular or moderator), or sit between the limbs that arise from the moderator band (muscular outlet). Not infrequently, different defects may be found in one heart, the condition called multiple VSDs (Fig. 2).
Figure 2. Ventricular septal defects as seen from the right ventricle. PM – perimembranous, JT – juxta-tricuspid, JA – juxta-arterial, Mi – muscular inlet, Mt – muscular trabecualr, Mo – muscular outlet (Freedom et al., 2004).
The conduction system follows the posteroinferior border of the perimembranous defect, the anterosuperior rim of the inlet defect and is not related to other types of VSDs.
Pathophysiology
A defect between the ventricles allows the blood to escape from the left ventricle into the pulmonary artery. The pulmonary blood flow becomes excessive and forces the pulmonary artery capillaries to contract. A pulmonary artery spasm is a normal condition for the newborns because the first month of living is a transitory period for breathing adaptation. The pulmonary resistance falls from the second month of age. As soon as the natural pulmonary resistance falls, the lungs are overcirculated. Blood return from the lungs loads the left ventricle and dilates this chamber. As a result, the baby develops heart failure that worsens from the third to fourth months. If left untreated, the pulmonary resistance will rise again. At this stage, the baby may even improve spontaneously because the left-to-right shunt diminishes. However, with time, the pulmonary resistance grows to extremely high level and the vasospasm becomes irreversible (Freedom et al., 2004; Ramaswamy, 2013; Stark, de Leval, Tsang, 2006). Irreversibly high pulmonary hypertension occurs in the course of a couple of years. In this condition, the lungs push off the blood from the right ventricle, so the righ-to-left shunt develops. The patient becomes blue and gets secondary polycythemia with aging. Condition, called Eisenmenger syndrome, is probably the only grim scenario nowadays for individuals with an untreated VSD (El-Chami, 2014).
The size and location of the defect determine its natural course. Small defects, usually a few millimeters in diameter, have very high probability of spontaneous closure by the age of 4 years and thus carry no risks. Moderate defects, a third of the diameter of the aorta, may lead to left ventricle overload and some degree of heart failure but typically carry low risks of Eisenmenger syndrome. Big VSDs, more that one half of the aorta’s diameter, are symptomatic and the most dangerous (Ramaswamy, 2013). Some defects, being initially moderate or large, partially close by involving surrounding tissues of the valves or valve chordae. These defects may be completely asymptomatic, but should an infectious agent occur in the blood stream, infectious endocarditis readily starts within the VSD. Infective endocarditis, if left untreated, is lethal (Brusch, 2014).
Presentation & Diagnostics
A VSD clinical course is dictated by its size. Small VSDs are asymptomatic – these children present slight systolic murmurs on the left sternal border. Big defects push patients into heart failure in the first moths of life. They gain weight poorly, develop excessive sweating, parents notice breathlessness and fatigue at feeding. Recurrent pulmonary infections are a common condition. Patients with Eisenmenger syndrome are cyanotic, complaint of exertional dyspnea, chest pains, and hemoptysis.
Physical findings include hyperactive precordium in symptomatic patients. Systolic murmur may be hard, but diminishes when pulmonary resistance rises significantly. In pulmonary hypertension, the P2 is split and the pulmonary component is accented. Signs of heart failure include moist lungs, hepatomegalia, and edema.
Echocardiography easliy visualizes the VSD. The presence of a defect is determined by signal absence in the interventricular septum (Fig. 3).
Invasive (angiography) and more sophisticated (computer tomography) techniques are used in complex cases.
Management
Individuals with small VSDs are asymptomatic, require no interventions, and undergo the protocol for infective endocarditis prevention (Wilson et al., 2007). Large defects cause early symptoms. Medical support with digoxin, angiotensin-converting enzyme inhibitors, and diuretics may alleviate heart failure symptoms, but surgery in unavoidable. A patch of pericardium or an artificial material is sewn into the defect under the cardiopulmonary bypass. The surgery carries some risks of conduction system damage, a surgical site infection, but usually is associated with low mortality or morbidity. Recent advances offer patients endovascular implantation of specially designed devices in anatomically suitable cases and usually bigger children. Moderate VSDs should be considered for elective closure on careful assessment of left ventricle load (Ramaswamy, 2013).
Conclusion
The ventricular septal defect is a hole between the ventricles of congenital origin. It is the most common congenital anomaly. Various classifications of VSDs have been proposed with regard to anatomy and surgical management. Diagnostics of the VSD is usually non-invasive. Conservative treatment of this disorder has only added importance. Moderate and large defects require timely interventions to prevent potentially lethal complications.